Blood Test for Cancer – An elegant concept
There is an elegance in the concept of a single and simple blood test being able to detect early stage cancer before symptoms and thereby enabling effective preventive treatment. This concept remains a very active area of research. However, in order to be clinically useful any such test must have sufficient sensitivity and specificity.
The sensitivity refers to the ability of a test to correctly identify those with the disease (true positive rate), whereas specificity is the ability of the test to correctly identify those without the disease (true negative rate). A good screening test should have both high sensitivity (to avoid missing those with a cancer), but equally have high specificity (to minimise those who would be erroneously said to have cancer with the anxiety and subsequent unnecessary investigations or even treatment that would induce).
For cancer detection there are 2 types of blood tests which can be potentially used – tumour markers and circulating tumour DNA
Tumour markers
Tumour markers have traditionally been proteins or other substances that are made by both normal and cancer cells but at higher amounts by cancer cells. These can be found in the blood, urine, stool, tumours, or other tissues or bodily fluids of some patients with cancer. Increasingly, however, genomic markers such as tumour gene mutations, patterns of tumour gene expression, and nongenetic changes in tumour DNA, are being used as tumour markers.
Tumour markers may be used for the following purposes:
- Screening for asymptomatic cancers although the necessary level of specificity and sensitivity has so far only been reached by a few tumour markers e.g prostate specific antigen (PSA). Even then, the UK NHS does not advocate the use of these for screening the general population.
- Monitoring of cancer survivors after treatment, and in the detection of recurrent disease.
- Diagnosis of specific tumour types, particularly in certain brain tumours and other instances where obtaining tissue biopsy is not feasible.
- Confirmation of diagnosis to verify the characteristics such as size and aggressiveness of a tumour and thereby to help in the evaluation of a suitable treatment schedule.
- Staging: some tumour markers are included in the staging procedures for some tumour localizations.
- Prognosis to plan the treatment when used pre-treatment and to help the patient to plan his future when used after the operation of cure.
- To verify the effect of treatment and to change the treatment if ineffective.
- As companion diagnostic to verify if the treatment is suited for the type or subtype of tumour particularly in personalized medicine.
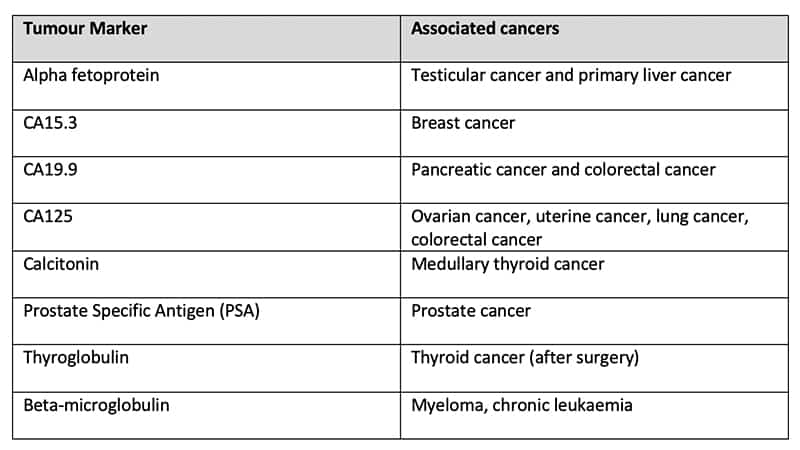
Examples of commonly used tumour markers include:
Circulating tumour DNA
Circulating tumour DNA (ctDNA) is found in the bloodstream and was first described in 1948. It refers to DNA that comes from cancerous cells and tumours. Most DNA is inside a cell’s nucleus. As a tumour grows, cells die and are replaced by new ones. The dead cells get broken down and their contents, including DNA, are released into the bloodstream. ctDNA are small pieces of DNA, usually comprising fewer than 200 building blocks (nucleotides) in length.
Over the last decade or so, there have been phenomenal technological advancements that have enabled scientists to detect and sequence ctDNA in the blood. As such, there is considerable excitement in the potential utility of ctDNA in cancer detection and treatment. The quantity of ctDNA varies among individuals and depends on the type of cancer, its location, and especially its stage.
Detection of ctDNA can be potentially helpful in the following applications:
- Prognosis determination – absence of ctDNA after surgery is associated with a much better prognosis and smaller chances of relapse. Prognosis determination aids in selecting the aggressiveness of treatment as well as determining the necessity for adjuvant therapy; patients at high risk of relapse could receive targeted treatment, while low risk patients would be spared unnecessary chemotherapy.
- Monitoring for treatment efficacy/relapse – Raised ctDNA concentrations or increased number of mutations indicate treatment failure/relapse earlier than clinical relapse. Conversely, a lack of ctDNA in the bloodstream indicates that the cancer has not returned.
- Selection of treatment – analyzing the genome of tumour cells using ctDNA can help doctors determine which specific treatment will be most effective.
- Tumour size/disease burden – larger amount of ctDNA in blood correlates with advanced tumour stage/greater metastatic burden. Its measurement could avoids the risk of ionising radiation from sequential CT scanning.
- Detection in asymptomatic individuals – this is the ‘holy grail’ of ctDNA and enormous sums of money are being ploughed into this area with attractive claims being made for its efficacy in detecting cancer.
Summary – will there be a universal blood test for cancer?
While recent studies demonstrate reasonable sensitivity and specificity for larger cancers, which are at a more advanced stage and secreting significant amounts of ctDNA, they are poor in detecting early stage tumours which are those one most wants to detect as this is when curative treatment would be most effective. For small cancers, there is generally just not enough ctDNA present to allow for an accurate test result. Further improvements in technology may allow for detection of even smaller amounts of DNA and if validated, would allow for early screening. However, at present, the reality does not match up to the hype and ctDNA is significantly inferior to imaging based means of cancer detection.